Technology
Our Approach
Our approach is based on the central premise that we can learn from patients who are winning their fight against cancer in order to treat those who are not.
Using our proprietary platform technologies, we are analyzing the T cells of cancer patients with exceptional responses to immunotherapy to discover clinically relevant targets and TCRs. We are building the ImmunoBank with the goal of delivering customized multiplexed TCR-T therapy to a wide range of patients with hematologic malignancies and solid tumors.
Learning
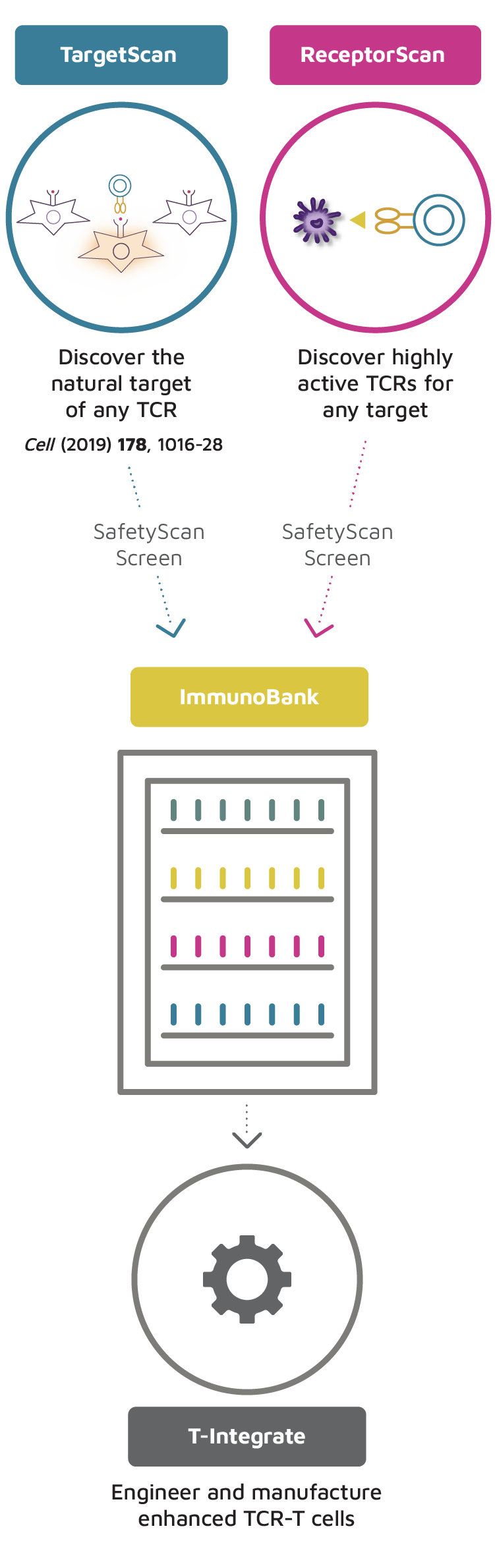
When a patient responds to an immunotherapy drug such as an immune checkpoint inhibitor, their tumor shrinks because T cells in their tumor become activated and drive an anti-tumor cytotoxic response. The TCRs of their T cells recognize tumor-specific antigens on tumor cells and signal the T cell to kill the cancer cells. Our approach starts with isolating clinically active anti-cancer T cells from tumor samples of patients who are actively responding to immunotherapy agents. We then use our proprietary TargetScan technology to determine the precise targets being recognized by their TCRs. This provides us with a novel TCR/target pair that can be developed into a TCR-T therapy candidate. In addition to discovering novel TCR/target pairs, we are leveraging our proprietary ReceptorScan technology to identify highly active TCRs against previously identified and clinically validated targets. Using SafetyScan, we then select TCRs that are highly active with no apparent problematic off-target effects to be added to our ImmunoBank.
Our Proprietary Target and TCR Discovery Process
Treating
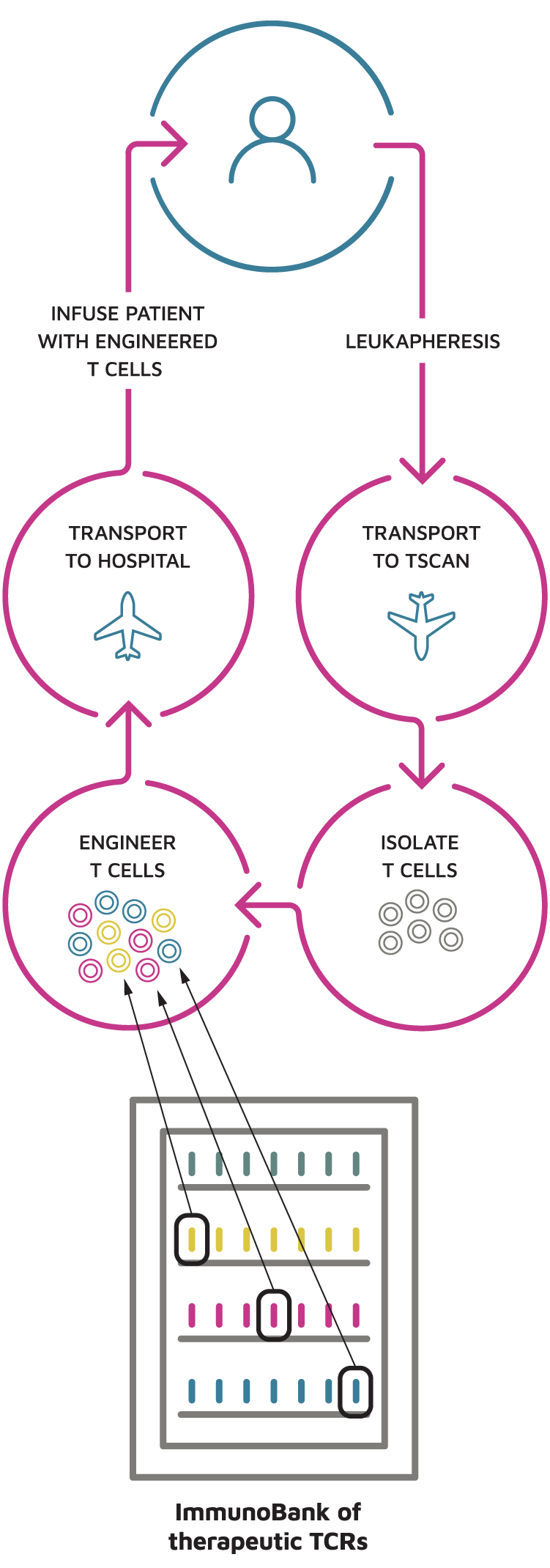
Our discovery process enables us to build and expand the ImmunoBank with what we believe represents the most active TCRs isolated from a large group of diverse patients who are responding to immunotherapy. We are developing TCR-T therapies that use these clinically relevant TCRs to reprogram the T cells of patients who do not spontaneously generate effective anti-cancer T cells and thus do not respond to immunotherapy.
Our Patient Treatment and Manufacturing Process
The TScan Advantage
- Our TCR-T therapies are based on highly active TCRs that are clinically relevant. Our platform analyzes anti-cancer T cells from a wide variety of patients in order to find the most active TCRs against each target. Many other approaches to T cell therapy rely on specifically expanding T cells that are already in the patient. By selecting the most active and clinically relevant TCR targets isolated from a broad group of patients who are responding to immunotherapy, we believe we can develop highly effective TCR-T therapies.
- Our TCR-T therapies are designed to be used in combination with each other. We are building our ImmunoBank of TCRs to allow for multiplexed TCR-T therapy, which has the potential to address a diverse array of solid tumors. We believe this approach may allow us to overcome the limitations and challenges of TCR-T therapy development to date including the heterogenous nature of solid tumors and overcoming resistance due to loss of a single target. We are populating the ImmunoBank with TCRs for multiple targets as well as multiple common HLA types for each target, thus helping us to overcome the key solid tumor resistance mechanisms of target loss as well as HLA loss.
- Our approach is expandable. ImmunoBank has the flexibility to be used with new and optimized methods of T cell engineering that we may develop over time. We are building the ImmunoBank to be compatible with both autologous and allogeneic engineering technologies in order to potentially transition to generating off-the-shelf, allogeneic T cells that have been pre-engineered with our TCRs for direct administration to patients.